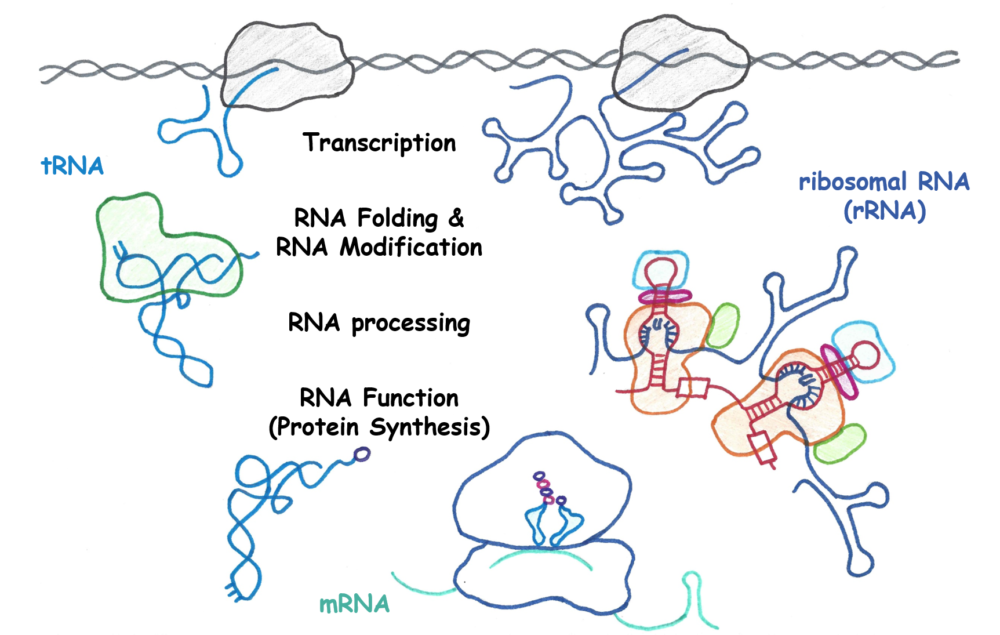
The Kothe research group studies the biogenesis and functions of RNA in gene expression as well as the dysfunctions of RNA in diseases such as cancer and inherited bone marrow failure syndromes and intellectual disabilities. Focussing on the role of chemical modifications of RNA, we have described an unexpected connection between the modification of RNA and the folding of RNA into its functional, three-dimensional shape. Building on this novel concept, we are in a unique position to investigate the mechanisms and functional consequences of RNA modification and folding as well as the connection to transcription and RNA processing for important RNAs such as transfer RNA (tRNA) and ribosomal RNA (rRNA). Our investigations will provide fundamental knowledge in RNA biochemistry, uncover molecular bases of inherited diseases, and unlock formation the cell’s protein synthesis machinery as a target for cancer treatment.
Ribosome Synthesis & H/ACA small Ribonucleoproteins
Ribosome biogenesis is a crucial process for each cell. In particular, fast dividing cells such as tumor cells have a high demand for ribosomes in order to rapidly produce all cellular proteins. Since ribosome biogenesis represents a bottleneck of the rapidly dividing cell, the pathway of ribosome biogenesis might be an additional target in cancer therapy. Therefore, our research aims at characterizing the critical steps in this crucial cellular process to identify new targets for tumor therapy.
During the early, co-transcriptional stages of eukaryotic ribosome synthesis, numerous pseudouridine are introduced in ribosomal RNA (rRNA) by H/ACA small nucleolar Ribonucleoproteins (snoRNPs) consisting of an H/ACA guide RNA as well as four proteins (Cbf5/Dyskerin, Nop10, Gar1, and L7Ae/Nhp2). Whereas the H/ACA guide RNA base-pairs with rRNA, the Cbf5/Dyskerin protein catalyzes the pseudouridylation reaction. We investigate the dynamic mechanism of action of H/ACA snoRNPs and their impact on the structure, folding and processing of ribosomal RNA.
Importantly, mutations in H/ACA protein genes cause an inherited bone marrow failure, Dyskeratosis congenita. Patients suffering from this inherited disease display a variety of symptoms including nail dystrophy, skin abnormalities, increased risk for cancer development and often early death before the age of 30. Our research studies the effects of mutations in H/ACA proteins on their function in ribosome biogenesis.
tRNA modification and function
tRNAs are the most highly modified RNAs in all cells. There is a huge diversity of different modifications, and each tRNA isoacceptor carries a unique set of several modifications. Recent research has uncovered how disease state and environmental conditions influence tRNA modification states which in turn affects tRNA function during protein synthesis. The Kothe group is studying the mechanisms and functions of different tRNA modifying enzymes with a special focus on understanding the interplay of different modifications, their impact on tRNA folding and the consequences for tRNA aminoacylation and protein translation.
Pseudouridine Synthases
In all domains of life, stand-alone pseudouridine synthases recognize specific RNA structures and sequences allowing them to site-specifically modify uridines to pseudouridines. Importantly, pseudouridine synthases modify all types of RNA including mRNA and long non-coding RNAs, but we often do not understand the functional consequences of pseudouridine formation and the RNA specificity of the pseudouridine synthases. We are investigating the mechanisms, specificity, and cellular functions of pseudouridine synthases in bacteria, yeast, and humans. Thereby, we will also gain insight into the role of these enzymes in various inherited diseases including brain development and function.
Methodology
The Kothe group applies a modern, multi-disciplinary approach of biochemistry, biophysics, molecular biology, and genetics together with transcriptomics. Only this combination of techniques will ultimately allow us to obtain a detailed understanding of RNA modification, folding and processing and formation of RNA-protein complexes such as the ribosome.
We recombinantly over-express proteins in E.coli and purify them by various chromatographic methods. To generate RNAs, we apply in vitro transcription followed by different purification techniques. Often, we apply molecular biology to clone the genes of interest and to modify them by site-directed mutagenesis. Thereby we can reconstitute active enzymes and ribonucleoprotein complexes in the test tube from highly purified components. This is the basis for further biochemical biophysical studies addressing the function of these complexes.
As biochemical assays, we use a variety of enzymatic assays as well as RNA-protein interaction studies including nitrocellulose filtration assays, electrophoretic mobility shift assays, and crosslinking. Furthermore, fluorescence measurements allow us to assess the structure, the dynamics, and the kinetics of RNA modifying enzymes and ribonucleoproteins. By rapid kinetic techniques, we can follow the time courses of RNA modification in real time.
Biochemical and biophysical measurements are complemented by molecular biology, genetics, and transcriptomics in the living yeast cell. Thereby, we can explore the cellular function of RNA modifications. Recently, we also began to conduct structural studies to visualize RNA-protein interactions.